Niels Bohr's theory of the atom tells us that electrons are not
:max_bytes(150000):strip_icc()/PeriodicTableCharge-BBG-58b5c80a3df78cdcd8bbb6c8.png)
In other words, the way in which an atom's electrons are arranged around its nucleus affects the properties of the atom. The "periodic" nature of chemical properties that Mendeleev had discovered is related to the electron configuration of the atoms of the elements. Correct! Electron configuration and the table Sodium is similar to lithium in terms of chemical properties. Elements in a given group in the periodic table share many similar chemical and physical properties.ī. Columns in the periodic table are called groups. Moves from left to right in a given period, the chemical properties of the elements slowly change. Rows in the periodic table are called periods. Thus sodium begins a new row in the periodic table and is placed directly beneath lithium, highlighting their chemical Sodium (Na, z = 11), however, is a silver metal that is solid at room temperature, much like the element lithium (z = 3).

The next element in order of atomic number is more similar (chemically speaking) to the first element in the row above it thus a new rowįor example, oxygen (O), fluorine (F), and neon (Ne) (z = 8, 9 and 10,respectively) all are stable nonmetals that are gases at room temperature. At the end of each row, a drastic shift occurs in chemical properties. As one moves from left to right in a row of the periodic table, the properties of the elements gradually change.
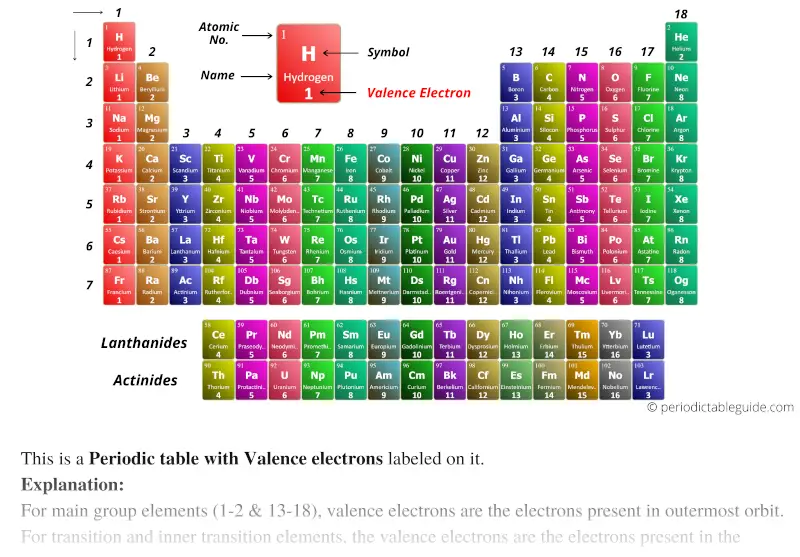
The modern periodic table of elements is based on Mendeleev's observations however, instead of being organized by atomic weight, the modern table is arranged by atomic number (z).
